About this course:
The purpose of this activity is to review the terminology regarding potential acts of bioterrorism, examine the potential effects, signs, and symptoms of biological, chemical, and nuclear/radioactive agents, the appropriate personal protective equipment (PPE), and evidence-based management of victims. This course also satisfies the nursing requirement for continuing education training on Bioterrorism in the state or Nevada.
Course preview
After this activity, the learner will be prepared to:
- define the terms terrorism, weapons of mass destruction (WMD), and bioterrorism
- discuss the utilization of syndromic surveillance and the Health Alert Network (HAN), including reporting procedures following an act of terrorism, and the proper utilization of standard precautions
- describe the most common examples of biological weapons, the signs and symptoms of exposure, appropriate corresponding PPE for medical personnel, and the evidence-based management of victims
- determine the most common examples of chemical weapons, the signs and symptoms of exposure, appropriate corresponding PPE for medical personnel, and the evidence-based management of victims
- recognize the most common examples of radioactive or nuclear weapons, the signs and symptoms of exposure, appropriate corresponding PPE for medical personnel, and the evidence-based management of victims
Terrorism is defined by the US Federal Bureau of Investigation (FBI, n.d.-a) as violent criminal acts committed by individuals or groups to further their ideological goals. If stemming from domestic influences (e.g., political, religious, social, racial, or environmental), it is defined as domestic terrorism. In contrast, foreign terrorism is inspired by or associated with a designated foreign terrorist organization or nation (FBI, n.d.-a).
Weapons of mass destruction (WMD) are destructive devices designed to cause death or serious injury through toxic or poisonous chemicals, a biological agent or toxin, or dangerous levels of radiation or radioactivity. These may be in the form of an explosive, bomb, rocket, or grenade (FBI, n.d.-b).
Bioterrorism is defined as the deliberate release of germs or other biological agents (e.g., bacteria, viruses, or toxins) sprayed into the air, used to infect animals or contaminate food or water supplies, or spread from person to person (Ready.gov, 2021).
In 2012, the Institute of Medicine (IOM) utilized a three-tier system for categorizing the care provided during a mass casualty event based on Hick and colleagues (2009) system as follows:
Conventional capacity indicates that the spaces, staff, and supplies used are consistent with daily practices within the institution. These spaces and practices are used during a major mass casualty incident that triggers activation of the facility’s emergency operations plan.
Contingency capacity indicates that the spaces, staff, and supplies used are not consistent with daily practices but provide care that is functionally equivalent to usual patient care. These spaces or practices may be used temporarily during a major mass casualty incident or on a more sustained basis during a disaster (when the demands of the incident exceed community resources).
Crisis capacity indicates that adaptive spaces, staff, and supplies that are not consistent with usual standards of care but provide sufficiency of care in the context of a catastrophic disaster (i.e., provide the best possible care to patients given the circumstances and resources available). Crisis capacity activation constitutes a significant adjustment to standards of care (IOM, 2012, Box 2-4).
The National Syndromic Surveillance Program (NSSP) is a timely system for detecting, understanding, and monitoring health events by tracking the symptoms of patients who present to emergency departments (EDs) and other care facilities in the US. This data collection allows public health officials to detect unusual symptom patterns. In addition, it provides an early warning mechanism for infectious outbreaks (e.g., COVID-19, influenza, Zika virus), national disasters, and other epidemics (e.g., the opioid epidemic, e-cigarette/vaping injuries). The NSSP is a cooperative effort by the Centers for Disease Control and Prevention (CDC) and federal, state, and local partners and health departments. This community of practice functions to electronically collect, share, and analyze data from patient encounters in EDs, urgent care facilities, inpatient healthcare centers, and laboratories via the BioSense platform. This collaboration includes over 6,000 facilities nationwide in 49 states and the District of Columbia and over 70% of EDs in the US. It was developed in response to the Public Health Security and Bioterrorism Preparedness and Response Act of 2002, Pub. L. No. 107-188. Within 24 hours of being entered into the electronic health record (EHR), the data is available for analysis by the NSSP. Facilities should establish multidisciplinary planning groups to establish procedures for detecting and appropriately reporting any potentially significant patterns within their institution, and healthcare professionals (HCPs) should be familiar with their facility’s protocols and local/state reporting mechanisms (CDC, 2021b).
The Health Area Network (HAN) is the CDC’s primary method of sharing urgent information with public health information officers, practitioners, clinicians, and laboratories. This collaboration of federal, state, territories, tribal, and local partners has developed a system to distribute public health information rapidly and efficiently. The HAN provides various message types, including health alerts, health advisories, health updates, and info services. Health alerts require urgent attention, as these are related to a specific incident and typically warrant immediate action by health officials, labs, clinicians, or the public. An example is a health alert describing cerebral venous sinus thrombosis cases with thrombocytopenia (a decreased platelet count) after receiving the Johnson & Johnson COVID-19 vaccine released on April 13 of 2021. Health advisories may not require immediate action but are related to a specific incident with associated recommendations or actionable items. An example was the warning regarding pneumonia of unknown etiology, which had infected 59 known patients in Wuhan, China, released on January 8 of 2020, leading to a level 1 travel advisory. Health updates are unlikely to require immediate action but provide additional information regarding a previously addressed topic. For example, on January 17, 2020, the HAN released a health update with interim guidance on the outbreak of the novel Coronavirus 2019 in Wuhan, China. This update reported cases had been identified in Japan and Thailand, community spread had not yet been confirmed, and the genetic sequence had been published. The HAN released additional health updates on the evolving pandemic on February 1, February 28, March 8, and again on March 15 of 2020. Finally, an info service distributes general public health information that is unlikely to require immediate action. All interested parties may add themselves to the HAN distribution list via the CDC’s website, and previous HAN messages are archived on the site for review on emergency.cdc.gov (CDC, 2020b).
HCPs also require adequate training and education regarding the proper management of mass casualty and bioterrorism attacks. The CDC has developed a training and education platform, the Clinician Outreach and Communication Activity (COCA), to prepare HCPs to respond to emerging public health threats and potential emergencies. COCA disseminates evidence-based health information, public health emergency messages and provides and promotes emergency preparedness and response training. This training is offered via conference calls, webinars, newsletters, live training events, and email support (CDC, 2018b). In addition, the CDC’s Healthcare Preparedness and Response Team (HPRT) has developed a digital toolbox with a PDF download containing tools and templates like customizable worksheets for organizational use. The CDC’s Hospital Preparedness Program (HPP) provides leadership and monetary grants to localities, territories, municipalities, and states to improve preparedness and enhance surge capacity for potential public health emergencies. The HPP is managed by the Assistant Secretary for Preparedness and
...purchase below to continue the course
PPE and Decontamination Basics
The US Occupational Safety and Health Administration (OSHA, 2005) defines first receivers as those HCPs who may care for victims at a local or regional hospital after being transported and triaged by first responders in the field in the event of a natural disaster or mass casualty/bioterrorism attack. There are numerous triage systems available currently. The Simple Triage Algorithm and Rapid Treatment (START) disaster triage system is based on the possibility of limited resources (supplies, medical personnel, medications, durable medical equipment) that need to be allocated and used to treat as many patients as possible. Disaster victims are initially triaged into one of four categories (Bhalla et al., 2015):
- Immediate (red triage tag) require significant treatment within 30-120 minutes. These patients have a respiratory rate greater than 30/min, require airway positioning to resume respiration, cannot follow verbal commands, or have no radial pulse combined with a capillary refill of greater than 2 seconds due to profuse bleeding.
- Delayed (yellow triage tag) require significant treatment, such as surgery, but can wait longer than 2 hours. They typically have a respiratory rate of less than 30/min, a radial pulse that is intact or a capillary refill under 2 seconds, and can follow verbal commands.
- Minor (green triage tag) include all walking wounded (minor injuries or illness).
- Expectant/deceased (black triage tag) is also referred to as expected to die. These victims are either deceased on arrival at the triage point or are severely injured and cannot be saved with the resources on hand without causing other casualties. This includes those that do not resume respirations after airway repositioning.
First receivers are at increased risk of potentially hazardous exposure to whatever biological, chemical, or radioactive agents were utilized in the attack. For this reason, OSHA recommends PPE use for first receivers working in hospital decontamination zones (HDZ, which includes any areas where the type and quantity of the hazardous substance are unknown and where contaminated victims, equipment, or waste may be present). Once the threat is identified, HCPs should utilize PPE appropriate for that particular substance. However, in the early stages of a mass casualty incident, before identification of the hazardous agent, the following PPE is recommended:
- a powered air-purifying respirator (PAPR) with a protection factor of 1,000 or greater
- a chemical-resistant protective garment (e.g., Tyvek protective suit)
- a head covering or hood, if not already included in the PAPR
- a double layer of protective gloves (butyl rubber may be more protective than nitrile for most [not all] toxic industrial chemicals and chemical warfare agents, but utilizing two gloves composed of two different materials may be ideal, or a foil-based glove)
- chemical-protective boots (OSHA, 2005)
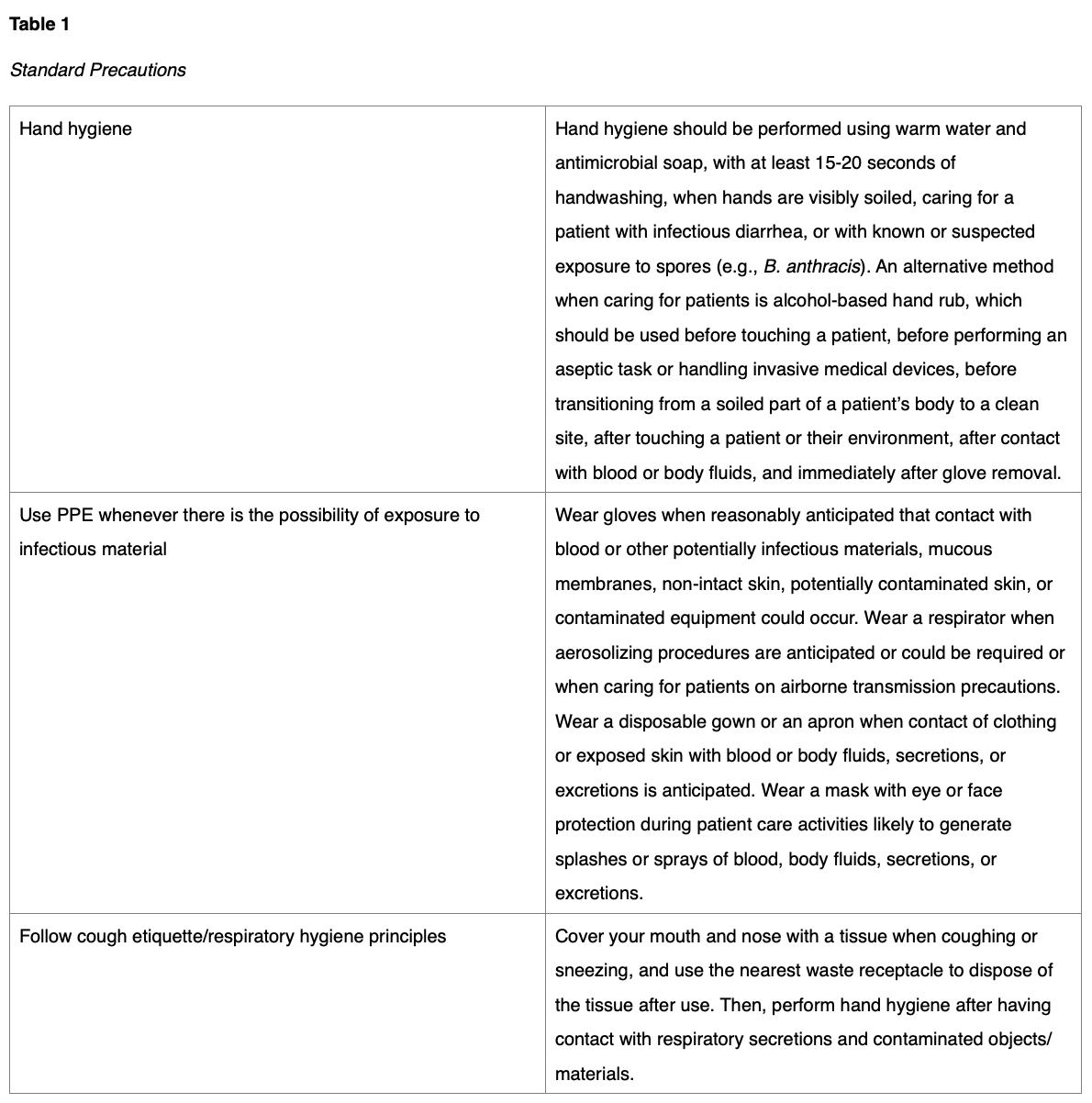
Removal of a victim’s clothing and decontamination before arrival at the hospital reduces contamination by 75%-90%. Clothing should be cut off using blunt-tip shears, and victims should be decontaminated using a shower of tepid water and mild liquid soap. HCPs should also be instructed to shower and decontaminate themselves after contact with potentially contaminated patients, equipment, or waste. HCPS working with victims that have been decontaminated (in the hospital post-decontamination zone) should be encouraged to utilize standard precautions (see Table 1) and standard PPE as indicated otherwise, such as disposable gowns, nitrile gloves, and surgical masks or N95 respirators if indicated. Outside of providing the appropriate PPE supplies, organizations and HCPs must ensure the proper use of these supplies to ensure their effectiveness, which means adequate training on PPE use, donning, doffing, and avoidance of accidental self-contamination (OSHA, 2005).


Biological Weapons
Category A
The CDC (2018a) categorizes potential biological hazards into categories A, B, and C. Category A agents pose a significant risk to national security because they can be easily spread to many people with a high mortality rate resulting in a significant public health impact, public panic, and social disruption. Therefore, these agents require special preparation from the public health sector. Examples of Category A agents include anthrax (Bacillus anthracis), botulism (Clostridium botulinum toxin), plague (Yersinia pestis), smallpox (variola major), tularemia (Francisella tularensis), and viral hemorrhagic fevers (e.g., Ebola, Lassa, or Marburg Fevers). Due to the elevated risk level, these agents will be discussed in greater detail (CDC, 2018a).
Anthrax
Anthrax is caused by the bacterium Bacillus anthracis (B. anthracis), a gram-positive, rod-shaped bacterium that lives predominantly in soil. It occasionally infects non-domesticated animals, leading to rare infections after contact with the infected animal or their byproducts. Otherwise, anthrax is not contagious unless the spores are ingested, inhaled, or contaminate a laceration or similar open wound. The spores are activated after entering the body, with signs and symptoms developing within 1-7 days if cutaneous or ingested and 7-60 days if inhaled. Cutaneous anthrax is fatal in 20% of cases if not treated, presenting with blisters, bumps, pruritis, erythema, and painless black ulcers most commonly found on the face, neck, arms, and hands. Ingested anthrax is fatal in about half of untreated cases and maintains a 40% mortality rate even with treatment (CDC, 2020a). When ingested or inhaled, B. anthracis creates two exotoxins responsible for its associated signs and symptoms: edema toxin (ET) and lethal toxin (LT). These toxins are constructed from edema factor (EF) and lethal factor (LF), which bind to a protective antigen (PA63) to create ET and LT and are then transported into target cells. Once within the cytoplasm, EF and LF alter cellular function, proliferation, and immune response (Bower et al., 2015).
Prevention of anthrax is possible via a US Food and Drug Administration (FDA) approved vaccine which is currently not recommended for the general US public. However, it is considered appropriate for certain lab personnel (who work closely with anthrax toxin or B. anthracis), members of the US military, and those who handle certain high-risk animals or animal products (e.g., veterinarians; CDC, 2016a). Post-exposure prophylaxis is recommended for anyone suspected of being exposed to anthrax using ciprofloxacin (Cipro) by mouth twice daily (PO BID). Common side effects from ciprofloxacin include nausea and vomiting (N/V), diarrhea, mild sunburn, and iatrogenic vaginal yeast infections. There is also an increased risk of tendinitis or tendon rupture and QT prolongation with ciprofloxacin use. An alternative is doxycycline (Vibramycin, Doryx) PO BID for adults. Either regimen should be continued for a total of 60 days of treatment. Common side effects from doxycycline include N/V, diarrhea, mild sunburn, and iatrogenic vaginal yeast infections (CDC, 2020a). The CDC also recommends three subcutaneous doses of the anthrax vaccine for previously unvaccinated adults if exposed to aerosolized B. anthracis spores (CDC, 2016a).
Symptoms of ingested anthrax infection include fever, chills, N/V, abdominal pain, and headache. Additional signs and symptoms may include cervical lymphadenopathy, pharyngitis, hoarseness, diarrhea, facial flushing, conjunctivitis, abdominal swelling, and fainting. Inhaled anthrax will also cause fever, chills, N/V, abdominal pain, and headache and is universally fatal if not treated. Even with treatment, inhaled anthrax infections carry a 45% mortality rate. Additional indications of inhaled anthrax include chest discomfort, shortness of breath (SOB), cough, confusion, dizziness, diaphoresis, fatigue, body aches, and tachycardia (CDC, 2020a). Meningitis is a potential complication of systemic anthrax. It typically presents with severe headaches, altered mental status, and meningismus (e.g., Kernig [pain with extension of the knee when the hip is flexed at 90º], Brudzinski [involuntary flexion of the hips/knees when the neck is passively flexed forward while lying supine], nuchal rigidity [neck stiffness], photophobia [light sensitivity], etc.). These patients may also develop other neurological signs or symptoms, such as seizures, cranial nerve deficits, papilledema, and limb weakness. In 2011, anthrax spores were used in 11 successful attacks against Americans, resulting in the death of 5 of the 11 targets. One victim developed meningitis, and all were infected via inhalation of anthrax spores (Bower et al., 2015).
Anthrax is diagnosed via blood cultures, which should be drawn before initiating antimicrobial treatment. Serum samples can also be tested for lethal toxin or using a real-time polymerase chain reaction (PCR) on either serum or cerebral spinal fluid samples. Sterile swabs of cutaneous or gastrointestinal (GI) lesions may also be tested (CDC, 2020a). An increase in transaminases is commonly seen in anthrax patient’s laboratory results, and chest imaging may indicate a pleural effusion or pulmonary infiltrate in patients exposed via inhalation. During periods of conventional treatment, a lumbar puncture (LP) is indicated if meningitis is suspected. However, in periods of contingency or crisis management, patients may be presumed to have meningitis without an LP if they exhibit 1 or 2 of the classic indicators of meningitis described above (Bower et al., 2015).
The existing anthrax treatment guidelines are based on minimal evidence. Pillai and colleagues (2015) found only 149 identified cases of systemic anthrax. Of these cases, 33 (22%) were complicated by meningitis, resulting in a 76% mortality rate amongst these 33 patients. They found an improved chance of survival in those treated with three or more parenteral antimicrobials (a combination of bactericidal and protein synthesis inhibitors [PSI] therapy) with a median duration of 14 days (Pillai et al., 2015). Bactericidal agents include beta-lactam agents (e.g., penicillins, imipenem [Primaxin], and meropenem [Merrem]), fluoroquinolones (e.g., ciprofloxacin [Cipro], levofloxacin [Levaquin], moxifloxacin [Avelox]), and vancomycin (Vancocin). Effective PSI agents include clindamycin (Cleocin), linezolid (Zyvox), doxycycline, chloramphenicol, or rifampin (Rifadin). Patients should be given 14 days of parenteral antibiotics (or until clinically stable) and then transitioned to oral medications. Those without signs or symptoms of meningitis can be managed using a bactericidal agent and a PSI. In those with confirmed meningitis (or awaiting confirmation via LP results), three antimicrobial agents are recommended during times of conventional care (Bower et al., 2015; Hendricks et al., 2014). See options in Table 2.

Monoclonal and polyclonal antitoxin therapy (e.g., Raxibacumab, Anthrax Immune Globulin [AIGIV]) is a potential adjunct to the antimicrobial treatment of anthrax. These antitoxins bind to PA63, thus blocking the intracellular transport of toxins. They are recommended for all patients with systemic anthrax in conventional care circumstances but are only recommended for severe sepsis/septic shock or those with meningitis within contingency or crisis care standards. During these periods, those with sepsis not defined as severe should be monitored closely for appropriateness. Any pleural fluid collections identified on imaging should be drained if feasible, and ascites should be drained using a Jackson-Pratt (JP drain) or ultrasound-guided (US-guided) paracentesis. Hemodynamically unstable pericardial effusions should also be drained (Bower et al., 2015).
Botulism
Botulism is caused by an infection of the gram-positive anaerobe Clostridium botulinum, which produces a neurotoxin. There are seven identified botulinum toxin serotypes, A-G. Most strains of C. botulinum produce just one serotype, ad serotypes A, B, E, and F cause most human morbidity and mortality (Rao et al., 2021). Botulism is a reportable/notifiable disease, meaning that the local/state health department should be notified of a suspected case. It is most often contracted via infected food. For example, spores can be found in honey, which is the reasoning behind the current recommendation against feeding honey to infants under 12 months old. Other risk factors include those who ingest home-canned or preserved food, drink home fermented alcohol, inject illicit drugs, or even obtain botulinum toxin injections for cosmetic or medical reasons (CDC, 2019a; Rao et al., 2021).
Signs and symptoms (Table 3) include facial paralysis due to prominent cranial nerve palsy. This may be evidenced by diplopia (double vision), dysphagia (difficulty swallowing), ptosis (drooping eyelids), dysarthria (slurred speech), and blurry vision. This typically flaccid paralysis progresses inferiorly (descending) and symmetrically, eventually affecting the arms, diaphragm, and legs, resulting in respiratory distress. The toxins affect motor function while sensory function remains intact (CDC, 2019a; Rao et al., 2021). This is due to the toxin’s underlying pathology, which inhibits the function of acetylcholine at motor neuron synapses. The toxins produced do not cross the blood-brain barrier, and therefore do not affect the central nervous system (CNS). Additional symptoms may include GI disturbances (if ingested orally), dry mouth, or SOB. Fever and altered mental status are rare and alternate explanations for the patient’s symptoms should be considered (Rao et al., 2021).

The patient’s serum, stool, or gastric fluid can be tested for botulism neurotoxin. Preliminary results may be available from toxin testing within 24-48 hours, but final results take 96 hours. Wound or stool samples (or even a food sample) can be cultured to assess for the presence of C. botulinum or one of its two closely related relatives, C. butyricum and C. baratil. Culture results take 2 to 3 weeks to complete and must be done through a state health department but are performed free of charge. Serum samples must be drawn before the administration of any antitoxin therapy and collected without anticoagulants. A sterile, nonbacteriostatic water enema may be used to collect a stool sample, but tap water and glycerin-based suppositories should be avoided. Gastric aspirates should be collected before antitoxin therapy. Food containers with remnants (unwashed) may also be sent for testing. Samples should be shipped with refrigerant to maintain a temperature range of 36° – 46° F (2° -8° C; Rao et al., 2021).
Treatment for botulism depends on the supply and demand of resources and follows the IOM standards of crisis care described earlier. Treatment is mainly supportive and may involve an advanced airway and mechanical ventilation to maintain respiration and oxygenation combined with close neurological and respiratory monitoring (Rao et al., 2021). Botulism antitoxin heptavalent (BAT) may be administered to halt the disease’s progress but will not reverse the existing deficits. BAT contains the equine immunoglobulin (Ig) from 7 serotypes (A-G). A vial of BAT should be diluted 1:10 in sterile saline and administered intravenously (IV) at 0.5 mL/min for the first 30 min, doubling the rate every 30 minutes until a maximum of 2 mL/min. Adults should receive an entire vial, which may contain variable volume. Side effects may include headache, nausea, pruritis/urticaria, fever, rash, chills, and edema. A hypersensitivity reaction (HSR) is a significant complication of BAT and typically presents with urticaria, erythema, pruritis, angioedema, bronchospasms, wheezing, cough, stridor, laryngeal edema, hypotension, and tachycardia. The infusion should be stopped immediately, and emergency care provided as indicated. A delayed allergic reaction to BAT, often termed serum sickness, may present 10-21 days following the infusion. This typically presents with fever, urticaria, maculopapular rash, myalgia, arthralgia, and lymphadenopathy. Delayed reactions generally are not life-threatening and can be treated symptomatically (FDA, 2018). A supply of BAT is contained within the Strategic National Stockpile, and therapy should be administered early in the disease process for maximum benefit (within 2 days; Rao et al., 2021). Alternately, BabyBIG is a human-derived botulism immunoglobulin for infant botulism. It contains antibodies for botulism serotypes A and B collected from pooled adult plasma samples. BabyBIG can be obtained through a request to the Infant Botulism Treatment and Prevention Program in California (California Department of Public Health, 2021). With current practice standards and conventional standards of care, botulism has less than a 5% mortality rate. The most commonly reported complications include deep vein thrombosis (DVT) or other thromboembolic events and ventilator-associated pneumonia (VAP), both of which can be prevented to a certain extent using diligent nursing care and chemoprophylaxis where indicated (Rao et al., 2021)
Plague
Plague is caused by the bacterium Yersinia pestis and is typically transmitted to humans through the bite of a rodent flea or after handling an ill animal. A plague epidemic in Europe in the Middle Ages killed millions, and a rat-associated outbreak occurred in urban Los Angeles in 1924-1925. Plague continues to infect humans regularly in Asia and Africa and rural areas of the western US in semi-arid upland forests and grasslands. Droplets can also spread the bacterium, especially if an ill individual with pneumonic plague has close contact with others while infectious and actively coughing. Depending on how the bacterium is encountered, plague can take bubonic, septicemic, or pneumonic form, among others. Bubonic plague results from the bite of an infected flea and leads to fever, headache, chills, weakness, and large, tender, swollen lymph nodes (buboes). Septicemic plague results from a flea bite or handling an infected animal and may develop independently or because of untreated bubonic plague. Symptoms include fever, chills, weakness, abdominal pain, shock, and bleeding. Septicemic plague may cause hemorrhage and necrosis of skin and other organs, especially distal limbs and appendages. Pneumonic plague develops after inhaling droplets or if bubonic or septicemic plague spreads to the lungs. It is characterized by fever, headache, weakness, SOB, cough, chest pain, and sometimes watery or bloody mucous. Pneumonic plague is the most severe form, leading to respiratory failure and shock if not properly treated, and is contagious from person to person via droplets. Patients with respiratory symptoms due to suspected plague should be isolated in a negative-pressure private room with airborne transmission precautions instituted. Post-exposure prophylaxis is recommended for those thought to be exposed to plague using oral doxycycline or ciprofloxacin (CDC, 2020d).
Patients suspected of having plague should be reported to local/state public health authorities. Diagnosis should be confirmed with blood, sputum, or lymph node aspirate cultures. Laboratory specimens should be drawn, and then treatment should be initiated in suspected plague patients while awaiting confirmation culture results. Treatment initiation should not be delayed. The presence of bipolar-staining, ovoid, Gram-negative bacteria with a safety pin appearance is presumptive for plague. Gentamicin IV daily can be used for adults. Adjunctive therapy may be required in those with bubonic plague. Fluoroquinolones may also be effective. IV antibiotics should be continued for 10-14 days or until the patient has been afebrile for 48 hours. The patient may be transitioned to oral therapy after improving clinically. Pediatrics can be managed with the same options, with dosing based on weight. Gentamicin should be given IV every 8 hours. Levofloxacin dosing for children should be IV every 12 hours or ciprofloxacin IV every 12 hours (CDC, 2020d).
Smallpox
Smallpox is a viral illness caused by the variola virus. Smallpox can be transmitted by droplets, making infected patients contagious if they have any active oropharyngeal lesions until all scabs slough off. Before eradication of the disease in 1980, it was associated with a 30% mortality rate. Care for patients with smallpox necessitates the use of standard precautions in addition to contact and airborne transmission-based precautions (i.e., disposable gown, gloves, N95 respirator, negative-pressure private room, and eye protection when indicated). The clinical definition is an acute viral illness with a fever above 101 °F followed by a rash with firm, deep-seated vesicles and pustules, all in the same stage of development. There are four clinical subtypes: standard (Variola Major), modified, flat, and hemorrhagic. Variola Major (VM) has an incubation period of 7-19 days (most 10-14 days) during which infected individuals are not contagious. The prodromal stage typically lasts 4 days and consists of fever, fatigue, prostration, headache, back pain, vomiting, abdominal pain, chills, anorexia, and pharyngitis. A rash may also be visible in patients with fair complexions. The eruptive stage consists of a resolution in the patient’s fever as a macular rash develops. The macules transition to papules and then vesicles over 4-5 days. The rash typically affects the oropharyngeal areas, face, and extremities, followed by the trunk, palms, and soles. The vesicles transition to pustules over 1-2 days, scabbing by day 9 and sloughing off by day 14. Scarring is possible, and corneal scarring may lead to vision loss. Other potential complications include encephalitis, osteomyelitis, pregnancy loss, and male infertility (CDC, 2019c).
There are currently two licensed vaccines for smallpox in the US. ACAM2000 is a replication-competent vaccine that contains the vaccinia virus (related to variola). It may cause a rash, fever, headache, and body aches. ACAM2000 is administered via multiple percutaneous punctures. It is currently recommended by the CDC for certain lab personnel at increased risk of occupational smallpox exposure and may be offered to specific HCPs at increased risk. This vaccine may also be effective if used as post-exposure prophylaxis in a crisis. Jynneos (Imvamune, Imvanex) is a replication-deficient vaccine containing attenuated live viruses, allowing it to be safely administered to immunocompromised patients. It is administered subcutaneously (SQ) via two doses separated by at least 4 weeks. Aventis Pasteur has an unlicensed investigational vaccine for smallpox (APSV) which is also replication-competent. It is administered via the same multi-puncture technique described above for the ACAM2000 vaccine, and a supply is included in the Strategic National Stockpile (SNS) as a backup option if the supply of ACAM2000 is insufficient (CDC, 2017b).
Modified smallpox occurs in vaccinated individuals. The prodrome typically lasts as long as in VM but consists of only a headache, fever, and back pain. Lesions are less numerous in modified smallpox, develop more superficially, and sloughing completes by day 10. Flat smallpox is rare, with increased toxemia and a higher mortality rate due to a deficient cellular immune response to the virus. It is more often seen in pediatric populations. Lesions develop more slowly, then merge but remain flat without forming pustules. Hemorrhagic smallpox is more often seen in adults, especially pregnant patients. This subtype is characterized by a shorter incubation period, increased symptoms during the prodromal stage, dusky erythema, profound toxemia, and multi-organ failure leading to death (CDC, 2019c).
The diagnostic criteria for smallpox are categorized into major and minor criteria. The three major criteria include:
- fever over 101° F that develops 1-4 days following a rash with associated weakness, headache, back pain, chills, vomiting, or abdominal pain (fever in addition to at least one other symptom)
- classic cutaneous lesions: firm, round, well-circumscribed, deep-seated vesicles or pustules
- lesions in the same developmental stage on any one body part
The five minor criteria include:
- lesions in a centrifugal distribution (greatest concentration on the face and distal extremities)
- initial lesions developing on the oral mucosa, palate, face, or forearms
- a patient who appears toxic or moribund
- lesions that progress from macules to papules to pustules over days
- lesions found on the palms or soles (CDC, 2019c)
A patient with all three major criteria is considered high risk for smallpox. They should be seen by a dermatologist and an infectious disease expert as soon as possible to confirm the criteria, the CDC should be contacted, digital photos taken, and the patient should be treated as clinically indicated without delay. A CDC Lab Response Network (LRN)-approved PCR test for the variola virus DNA should be initiated to confirm the diagnosis. No other lab testing is required until smallpox has been ruled out (CDC, 2019c).
Moderate risk is defined as a patient who presents with a febrile prodrome and one additional major criterion or four of the minor criteria. They should be seen by a dermatologist and/or an infectious disease expert as soon as possible to confirm the criteria. Still, lab testing should rule out other differential diagnoses, such as varicella and other more common viral exanthems. Treatment should be initiated as clinically indicated for the most likely etiology (CDC, 2019c).
The CDC defines low risk as those patients without a febrile prodrome or those with a febrile prodrome in combination with one to three minor criteria. Low-risk patients should be tested for varicella and treated as clinically indicated. In addition to varicella, herpes zoster, impetigo, drug eruptions, contact dermatitis, erythema multiforme minor or major, enterovirus (i.e., hand, foot, and mouth disease), herpes simplex, scabies, molluscum contagiosum, and monkeypox are all vesicular rashes that should be considered (CDC, 2019c).
Treatment for smallpox is predominantly supportive, similar to other viral illnesses. Antipyretics, IV fluids, nutritional support, pain management, and symptomatic measures to alleviate the discomfort associated with the rash are all appropriate. Three antiviral medications have shown effectiveness in animal and in-vitro studies. However, due to the rare nature of smallpox, these drugs have not undergone clinical trials in human subjects. Tecovirimat (Tpoxx) is the only FDA-approved medication for the treatment of smallpox based on limited efficacy data. Cidofovir and brincidofovir are not currently FDA-approved. In-vitro and animal studies exist, but an emergency use authorization (EUA) or an investigational new drug (IND) protocol would be required from the FDA to allow their widespread use in a mass casualty response. A supply of both tecovirimat and cidofovir are included in the SNS (CDC, 2019c).
Tularemia
Tularemia is caused by the bacterium F. tularensis. It is most commonly found in rodents, rabbits, and hares. The bacteria may be aerosolized in a mass casualty attack, although most cases were historically transmitted via infected flea or tick bites. It is not known to be contagious, and thus transmission-based precautions are not required. The incubation period varies from 1-21 days, but the average is 3-5 days. Symptoms of exposure typically include fever, chills, headache, malaise, anorexia, vomiting, diarrhea, myalgia, arthralgia, and weakness. If individuals eat or drink contaminated food or water, this may lead to exudative pharyngitis with cervical, periparotid, or retropharyngeal lymphadenopathy. If inhaled, it often causes chest pain, cough, and hilar adenopathy, infiltrate, or pleural effusion on chest imaging. PCR and direct immunofluorescence assays (DFA) are available to support a suspected diagnosis of tularemia, as well as serum antibody titers. Cultures can confirm the presence of F. tularensis based on a clinical specimen (e.g., swabs or scrapings of ulcers, lymph node aspirates or biopsies, pharyngeal swabs, or respiratory specimens/pleural fluid), but labs should be warned of the increased risk associated with this bacterium. Cultures may require an extended incubation period due to the slow-growing nature of this bacterium. Serum samples collected during the acute illness phase and then 2-3 weeks later may also confirm the diagnosis based on seroconversion from IgM to IgG antibodies (CDC, 2021c).
Streptomycin is effective at treating tularemia but is not universally available in the US. Other antibiotics effective at treating tularemia include gentamicin. Pediatric patients should be dosed based on their weight. Ciprofloxacin (Cipro) may also be used. Both of these are preferred over doxycycline, which may be given IV or PO BID for 14-21 days in adults only. If available, chloramphenicol has been shown to increase the effectiveness of streptomycin in the treatment of tularemia meningitis (CDC, 2021c).
Viral Hemorrhagic Fevers
The viruses that cause these illnesses are RNA viruses enveloped in a lipid coating. Arenaviruses are found naturally in animals, especially rodents, and some cause viral hemorrhagic fever (VHF) in humans. Lymphocytic choriomeningitis virus (LCMV) causes Lymphocytic choriomeningitis, the Junin virus causes Argentine HF, the Machupo virus causes Bolivian HF, the Lassa virus causes Lassa fever, and the Lujo virus causes Lujo HF. Some viruses can spread person-to-person, including Lassa, Machupo, and Lujo, via exposure to an infected individual’s blood or body fluids. They are not spread via casual contact (CDC, 2018a).
Appropriate PPE when caring for unstable patients (i.e., those with active bleeding, vomiting, or diarrhea) with suspected or confirmed VHF includes a respirator mask, double gloves, impervious disposable gown, disposable boot covers, and goggles or similar eye and face protection (e.g., face shield). PPE should be donned and doffed in private, separate rooms with access to shower facilities. Access to patient rooms should be restricted to essential personnel only. All HCPs should be trained in the proper procedure for donning and doffing PPE using technology-based simulation or rehearsal-style training. The donning and doffing PPE process should be supervised by a trained observer wearing a fluid-resistant gown, face shield, surgical mask, double gloves, and shoe covers. Surgical scrubs or disposable garments should be worn under the recommended PPE, along with dedicated washable footwear. Alcohol rub should be utilized frequently and accessible during care provision and within the PPE staging area to sanitize gloved hands. Double gloving provides the opportunity to remove gross contamination when doffing PPE. While not airborne, the CDC recommends that HCPs utilize an N95 or greater respirator in case of a sudden need to perform an aerosol-generating care procedure, which is often an unanticipated and urgent need. The N95 should be worn with a full-face mask and/or a surgical hood if available. Alternatives include a PAPR with a full-face covering and head shroud, which decreases the risk of accidental self-contamination by the HCP but is also cumbersome and challenging to don/doff. A disposable apron is also recommended when caring for patients with active vomiting or diarrhea. The CDC offers a PPE calculator tool for organizations to estimate the amount of PPE supplies required to care for VHF patients (CDC, 2018d). The process for donning/doffing PPE can be found below in Box 1.

PPE recommendations when VHF patients are clinically stable (no active bleeding, vomiting, or diarrhea) include reducing the requirement for the disposable gown from impervious to fluid-resistant. A surgical facemask may be used in combination with a full-face shield (in lieu of an N95 respirator or PAPR). Double gloving is still recommended, as are surgical scrubs or disposable garments under the PPE. PPE should be inspected before donning, and hand hygiene performed. Gloves should be donned first, followed by the disposable gown, surgical mask, second pair of gloves, and face shield. As discussed above, PPE should be verified by an observer before entering the care environment. PPE should be inspected for visible contamination before removing, and gloves should be sanitized using an alcohol-based rub. Outer gloves should be removed first, followed by the inspection and disinfection of the inner gloves. The face shield should be removed next, followed by the disposable gown, disinfecting the gloves between these steps. The gloves should then be disinfected and changed before removing the surgical mask. The gloves can be disinfected a final time before removing them. The HCP should then perform hand hygiene and inspect their scrubs/undergarments for visible contamination (CDC, 2018c).
Lassa fever has a 1–3-week incubation period before patients become symptomatic. Roughly 80% of infected individuals develop mild disease, with characteristic fever, malaise, and headache. The remaining 20% develop mucosal hemorrhage, respiratory distress, vomiting, facial swelling, and chest/abdominal/back pain. Hearing loss is also common and may be permanent. Lassa fever is typically diagnosed via a positive enzyme-linked immunosorbent serum assay (ELISA) to detect IgM or IgG antibodies. A reverse-transcriptase PCR (RT-PCR) assay for the Lassa antigen can be utilized for confirmation if testing is done earlier in the disease process. A viral culture may also indicate the Lassa virus in 7-10 days. Treatment for VHF, including Lassa, is predominantly supportive. It often requires close monitoring, antipyretics to treat fever, IV fluids to maintain fluid and electrolyte balance, and may also require medication for pain or N/V, supplemental oxygen, vasopressors, nutritional support, and blood product transfusion if blood losses are substantial. Ribavirin (Ribasphere) is an antiviral medication that may prove beneficial if started early in the disease process. (CDC, 2019b). Ribavirin is a nucleoside analogue that inhibits protein synthesis. Common side effects include cough, constipation, heartburn, fatigue, headache, fever, arthralgia/myalgia, irritability, insomnia, N/V, and anorexia. It may cause anemia, neutropenia, and thrombocytopenia. Ribavirin is most commonly used to treat hepatitis C infection (Medscape, n.d.).
Members of a related genus of viruses, the Ebolavirus genus, may also cause VHF. This family is most notable for the Ebola virus disease caused by the Zaire ebolavirus and includes Sudan virus, Tai Forest virus, Bundibugyo virus, Reston virus, and the Bombail virus. Like Lassa, Ebola is a VHF with an incubation period of 2-21 days (mean 9-11). It is contracted through an infected individual’s blood/body fluids (including semen) if in direct contact with broken skin or a mucous membrane or through infected animals (e.g., fruit bats, primates). Patients infected with Ebola typically present with GI symptoms initially (around day 5), reporting N/V, abdominal pain, severe watery diarrhea, and anorexia. They may also develop chest pain, SOB, headache, confusion, conjunctival injection, hiccups, and seizures. Between days 8-12, they typically develop fever, chills, malaise, and myalgias, along with bleeding (e.g., ecchymosis, bruising, petechiae, or oozing of wounds/lacerations). Laboratory testing typically indicates leukopenia (i.e., a reduced number of white blood cells [WBCs]) and lymphopenia (i.e., a reduced number of lymphocytes, a type of agranulocytic WBCs) initially, followed by neutrophilia (i.e., an increase in neutrophils, a variety of granulocyte WBCs), a left shift (i.e., an increase in the number of bands, or immature WBCs), and thrombocytopenia (i.e., platelet count of < 50-100,000/µL, or 50-100 x 109/L). A PUI (i.e., a person under investigation) that is identified based on symptoms and potential exposure (epidemiological risk factors in the last 21 days) should be isolated in a private room with the door closed (and access to a private bathroom) and managed as if they are infected until proven otherwise. The local health department should be contacted immediately, which will work with the CDC to confirm the PUI status and give further instructions. Presumptive testing using real-time RT-PCR to detect the virus is available at 60 LRN facilities nationwide. A 4 mL serum sample is required, preserved with EDTA. The sample should be collected in a plastic test tube, triple packaged, and shipped with refrigerant to maintain a temperature range of 36° – 46° F (2° -8° C; CDC, 2021a).
The prevention of Ebola virus disease may be achieved using Ervebo, a live, attenuated recombinant vaccine. The CDC currently recommends the vaccine for certain adults at increased risk, such as employees of level 4 lab facilities and HCPs within federally designated Ebola treatment facilities. It is also in the SNS to be used in response to an outbreak. Supportive treatment for Ebola patients includes fluid resuscitation in response to hypovolemia and any electrolyte abnormalities, vasopressors to maintain adequate perfusion, supplemental oxygen to address any hypoxia, pain control, and nutritional support. Antimicrobials may be indicated for secondary infections, and septic patients should be treated according to the Surviving Sepsis guidelines. In addition to supportive treatment, there are two FDA-approved antiviral medications for managing Ebola virus infection (CDC, 2021a). Inmazeb is a combination of atoltivimab, maftivimab, and odesivimab-ebgn. The three monoclonal antibodies bind to the virus glycoprotein, blocking its entry into host cells. It was first approved in October of 2020. The most common symptoms reported by drug recipients include fever, chills, tachycardia, tachypnea, and vomiting (all symptoms commonly seen in patients with the Ebola virus; FDA, 2020a). Ansuvimab-zykl (Ebanga) is a single monoclonal antibody approved later in 2020 by the FDA. Like Inmazeb, recipients of Ebanga most often reported fever, tachycardia, diarrhea, vomiting, hypotension, tachypnea, and chills (FDA, 2020b). HSRs are possible, as with other antibody-based immunotherapy, and the infusion should be stopped immediately if this is suspected (FDA, 2020a, 2020b).
Categories B and C
Briefly, Category B agents are moderately easy to transmit and may result in significant morbidity but low mortality. These agents still require enhanced diagnostic capacity and disease surveillance through the CDC and its public health partners. Examples of Category B agents include:
- brucellosis (Brucella species)
- epsilon toxin of Clostridium perfringens
- food safety threats (Salmonella species, Escherichia coli O157:H7, Shigella)
- glanders (Burkholderia mallei)
- melioidosis (Burkholderia pseudomallei)
- psittacosis (Chlamydia psittaci)
- Q fever (Coxiella burnetii)
- ricin toxin from Ricinus communis (castor beans)
- staphylococcal enterotoxin B
- typhus fever (Rickettsia prowazekii)
- viral encephalitis caused by alphaviruses (e.g., eastern equine encephalitis, Venezuelan equine encephalitis, and western equine encephalitis])
- water safety threats (e.g., Vibrio cholerae, Cryptosporidium parvum; CDC, 2018a)
The CDC defines Category C agents as emerging pathogens with the potential for mass dissemination if adequately engineered due to their extensive availability and the relative ease with which they can be produced and then disseminated. If utilized in this manner, these agents maintain the potential for significant public health impact through their high morbidity and mortality rate. Examples within this category include the Nipah virus and the hantavirus (CDC, 2018a).
Melioidosis and Glanders
Caused by B. pseudomallei and B. mallei, melioidosis and glanders are closely related and treated similarly. Glanders is primarily a diagnosis of horses, mules, and donkeys. Signs and symptoms include fever, chills, muscle aches, chest pain, muscle tightness, headache, nasal discharge, and photophobia. Infections may be localized, pulmonary (if aerosolized), systemic, or chronic. Pulmonary infections lead to pneumonia and may result in a pulmonary abscess or pleural effusion. Chest imaging will often indicate a localized lobar infection (CDC, 2017a).
Melioidosis has an incubation period that ranges from 1-21 days but averages 9 days. Reports of latent infections becoming symptomatic years later are rare. Infection typically occurs after inhaling contaminated dust or droplets leading to mild bronchitis or pneumonia with associated fever, headache, cough, anorexia, chest pain, and muscle aches. It is typically diagnosed based on a urine, serum, sputum, skin/abscess culture. Chest imaging typically indicates cavitary lesions similar to tuberculosis (TB; CDC, 2016b).
Post-exposure prophylaxis for glanders or melioidosis can be accomplished using oral trimethoprim/sulfamethoxazole (Bactrim) or amoxicillin/clavulanic acid (Augmentin). Ill patients should be isolated, with diligent adherence to standard and airborne precautions, including the use of a negative-pressure private room and N95 respirators. For confirmed cases of glanders or melioidosis, ceftazidime (Fortaz) or meropenem (Merrem) can be infused every 8 hours for 10-14 days. This should be followed by 3-6 months of oral trimethoprim/sulfamethoxazole or amoxicillin/clavulanic acid. Of these, trimethoprim/sulfamethoxazole is the preferred treatment. Trimethoprim/sulfamethoxazole may lead to a severe allergic reaction (Stevens-Johnson Syndrome), bone marrow suppression, renal failure, liver dysfunction, hyperkalemia, increased creatinine. A complete blood count (CBC) and basic metabolic profile (BMP) should be checked weekly for the first three weeks of treatment and biweekly after that. Patients should also be given a folate supplement for the duration of treatment. Pregnant patients should be managed with amoxicillin/clavulanic acid (Lipsitz et al., 2012).
Ricin Toxin
Ricin toxin is derived from castor beans and is a waste byproduct of castor oil production. Ricin is lethal if injected, although this method is not feasible as bioterrorism or large-scale attack. Ricin is not absorbed by the skin but may be toxic if inhaled or ingested in substantial doses (National Academy of Sciences, 2004). Initial symptoms are likely to occur 4-8 hours following ingestion or inhalation, the most likely routes to be utilized in a mass casualty event. Ingestion of ricin may lead to hallucinations, hematuria, hypotension, weakness, flu-like symptoms (i.e., fever, myalgia, and arthralgia), profuse diarrhea (may be bloody), profuse vomiting, seizures, severe dehydration, and hypovolemic shock with multi-organ failure. While data on the effects of ricin inhalation are limited, symptoms may include cough, respiratory distress/bronchoconstriction, cyanosis, diaphoresis, hypotension, flu-like symptoms (i.e., fever, myalgia, and arthralgia), nausea, pulmonary edema, weakness, and respiratory failure with multi-organ failure. Repeated inhalation exposures may lead to an allergic-like response, with hives, urticaria, lacrimation, etc. Laboratory findings may include metabolic acidosis, elevated liver function tests (LFTs), renal function tests, hematuria, and leukocytosis (elevated WBC count). Although there are environmental tests to detect ricin used by certain government agencies, including the US Postal Service, specific diagnostic testing is limited. Biological tests for ricinine, an alkaloidal component of the castor bean plant, and PCR testing to detect castor bean DNA are available (CDC, 2018e).
Treatment for ricin exposure is supportive, as no antidote exists. Patients should be evacuated from the area of release and clothing removed. If clinically stable, decontaminate with mild soap and tepid water and copious rinsing. If inhaled, patients should be placed in the semi-Fowlers position with supplemental oxygen. Cardiopulmonary resuscitation and advanced airway support may be required. Pulmonary hygiene/toileting may also be beneficial (e.g., relaxed deep breathing, huffing, suction, spirometry, percussion). If ingested, activated charcoal should be administered, but induced vomiting is not indicated. If ingestion occurred within an hour, gastric lavage might be beneficial. If vomiting, position the patient to maintain the airway and prevent aspiration. IV fluid and electrolyte balance should be maintained using fluid resuscitation/replacement. IV pressors may be required if patients are severely hypotensive. Eyes should be flushed with tepid water for at least 15 minutes if ocular exposure. Antiepileptic drugs may be necessary to control seizure activity (CDC, 2018e).
Chemical Agents
Several chemical agents may be inadvertently or intentionally released into the environment, causing significant and widespread public exposure. This type of agent was first used during World War I. Gases may be disseminated through ventilation systems, aerosolized over a large, populated area, placed into explosive devices, used to sabotage industrial plants or vehicles, or used to contaminate food or water supplies. Many people (or animals) in one geographic area with corresponding signs and symptoms simultaneously may indicate a chemical attack (National Academy of Sciences, 2004). Potential chemical agents and their associated toxidromes will be outlined and briefly discussed. The US Department of Health and Human Services defines levels of PPE required in hazardous situations where exposure to potentially dangerous chemicals or radiation is a concern. Level B is required as a minimum level of protection upon initial entry into a site with unknown exposure. In addition, all respirators and breathing apparatus must be approved for use by the National Institute of Occupational Safety and Health (NIOSH; Chemical Hazards Emergency Medical Management [CHEMM], 2021d). These levels are outlined in Table 4.

Identifying a chemical agent is key to its management. The CHEMM has developed a prototype Intelligent Syndromes Tool (CHEMM-IST 2.0) on their website to help HCPs managing chemical exposure victims identify the most likely agent based on the patient’s signs and symptoms. The tool will allow users to insert the patient’s vital signs, mental status, pupil size, skin color/moisture assessment, and the presence of mucous membrane irritation, wheezing or crackles, and burns. The tool is currently in beta-testing and is not yet validated for widespread clinical use and only includes inhaled chemical agents at this time (CHEMM, 2021a).
Nerve Agents
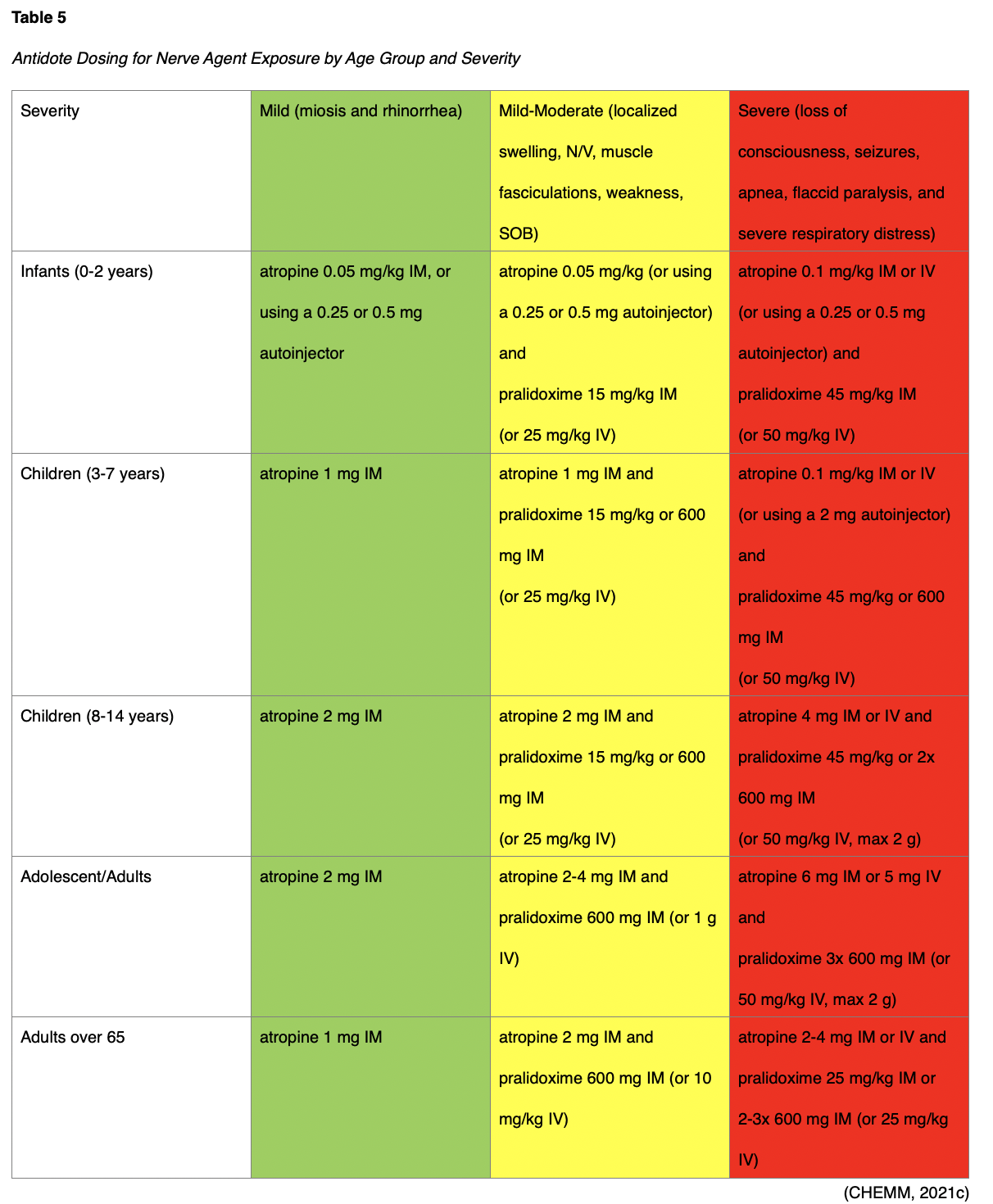
Nerve agents include sarin or VX. These odorless agents typically create headache, miosis (pinpoint pupils), rhinorrhea (nasal discharge), hypersalivation, localized swelling, difficulty breathing, chest tightness, SOB, seizures, weakness, and N/V. Sarin is one of the most toxic chemicals, becoming lethal at just 36 parts per million (ppm). Children are much more susceptible to nerve agents than adults. Fortunately, it is non-persistent, lingering in the environment for just minutes to hours after being dispersed. A delayed reaction to skin exposure may present 18 hours following exposure. Patients should be removed from the immediate area, clothing removed, and decontaminated with soap and water. Intramuscular (IM) atropine (AtroPen) and IM or IV pralidoxime chloride (2-PAM Cl) may be used (see Table 5), as well as IM or IV benzodiazepines for seizure management (CHEMM, 2021b).
Atropine ophthalmic drops may be used to treat miosis alone without other respiratory or systemic signs and symptoms. If administered IV, pralidoxime should be infused over 20-30 minutes. The use of an autoinjector is preferred over a traditional IM injection, as this decreases the time to therapeutic effect from 20-25 minutes to 8 minutes. The Mark 1 kit includes an autoinjector containing 600 mg of pralidoxime and one containing 2 mg atropine. The Duodote kit has a single autoinjector with 600 mg pralidoxime and 2 mg atropine. Patients who lose consciousness and become apneic after nerve agent vapor exposure may require up to 15 mg of atropine to regain consciousness. If symptoms are moderate, repeat doses of atropine should be administered every 5-10 minutes until dyspnea, ventilation resistance, and secretions decrease. Pralidoxime may be repeated, up to 45 mg/kg during the first hour. Atropine may be repeated every 2-5 minutes if symptoms are severe. Pralidoxime should be started IV is clinically possible and can be repeated hourly x 2 if symptoms are severe. Hypoxia should be corrected before atropine administration due to a risk of ventricular fibrillation (seen with atropine administration in hypoxic animal models), but atropine administration should not be delayed for this reason (CHEMM, 2021c; National Academy of Sciences, 2004).
Diazepam (Valium) is FDA-approved for treating seizures related to nerve gas exposure, although midazolam (Versed) is also used for this indication off-label. Of note, midazolam should be administered with caution in pregnant patients, as there is evidence of fetal risk (CHEMM, 2021c).
Blood or Systemic Agents
Blood or systemic agents cause knockdown syndrome, with a rapid loss of consciousness, seizures, hypotension, and cardiac arrest. Examples include hydrogen cyanide or sulfide, carbon monoxide, and arsine. These agents disrupt intracellular oxygen utilization or delivery. Hydrogen cyanide is highly toxic, becoming lethal at just 120 ppm. As a comparison, carbon monoxide becomes lethal at 4,000 ppm. The initial presentation of these patients may include fatigue, lightheadedness, and flushing. Victims should be immediately decontaminated, removing outer clothing, and thoroughly washing the hair. In most circumstances, level B or C PPE is indicated for HCPs, although level A may be required for some of these agents. Treatment should begin with supportive care, including supplemental oxygen and fluid resuscitation. If ingested, activated charcoal administration is indicated. Kits for the treatment of hydrogen cyanide exposure, referred to as cyano kits, typically contain amyl nitrite perles and IV solutions of sodium nitrite and sodium thiosulfate. The amyl nitrite perle should be broken into sterile gauze and applied under the patient’s nose. Patients should be monitored closely for hypotension with a reduction in the infusion rate if this develops. Alternative treatments include the use of hydroxocobalamin (CHEMM, 2021b; National Academy of Sciences, 2004).
For heavy-metal agents such as arsine, the gas form of arsenic, chelators may help to bind the heavy metals and reduce intoxication (CHEMM, 2021b). Dimercaprol (British anti-lewisite, BAL) was initially developed in wartime Britain for the treatment of heavy metal intoxication, followed by unithiol (DMPS) and succimer (DMSA), which are dithiol water-soluble analogs of BAL developed in the 1950s. DMPS and DMSA remain the treatment of choice for arsenic or inorganic mercury salt poisoning. They must be administered as soon as possible following exposure (within minutes to hours). BAL must be administered IM, while DMPS and DMSA both allow for oral or IV administration. BAL has also been found to increase the distribution of heavy metals to the brain, so its use is no longer recommended. Allergic skin reactions occur in up to 10% of patients, as well as GI symptoms, an increase in hepatic transaminases, or a mild reversible leukopenia (Kosnett, 2013).
Blister Agents
Blister agents, mustard agents, and vesicants are irritants and corrosives that primarily affect the skin, eyes, and respiratory tract. Examples include lewisite and sulfur mustard. Typical presenting signs and symptoms develop gradually and often include dyspnea, impaired vision, and skin lesions. Eventually, a pruritic rash followed by blistering may evolve. Other symptoms may take 2-4 hours to develop and include pharyngitis, dry cough, pulmonary edema, eye damage, N/V, and diarrhea. Patients should be removed from the immediate area. Clothing should be removed, and patients immediately decontaminated with soap and copious amounts of water. In most circumstances, level B or C PPE is indicated to protect HCPs, although level A PPE may be required for some of these agents. Supportive patient care is indicated, but no antidotes to these agents currently exist (CHEMM, 2021b).
Anticholinergic Agents
Chemical agents that block or otherwise inhibit the cholinergic receptors for acetylcholine include 3-quinuclidinyl benzilate (BZ) and other glycolate anticholinergics (e.g., atropine, hyoscyamine). These agents cause anticholinergic toxidrome, which typically presents with mydriasis (dilated pupils), anhidrosis (decreased sweating), elevated body temperature, tachycardia, mental status changes, and hallucinations. Patients should be removed from the immediate area, clothing removed, and decontaminated with soap and water. Anticholinergic toxidrome is typically treated using an approved antidote, such as physostigmine. Supportive treatments may include therapeutic cooling, benzodiazepines, and other symptomatic treatments as indicated (CHEMM, 2021b).
Radiation/Radioactive Agents
The US Department of Health and Human Services contains the Radiation Emergency Medical Management (REMM, 2021c). The REMM provides information for HCPs, a scarce resource triage tool, and hospital order templates. This group defines radiation exposure as when the whole or part of the body absorbs penetrating ionizing radiation from an external source, or a radionuclide is ingested, inhaled, or absorbed. They define contamination as when a radioisotope is ingested, inhaled, or deposited on someone’s surface (or clothing) in gas, liquid, or solid form after being released into the air. The primary difference between these two terms is that exposed individuals cannot expose others, yet a contaminated individual can expose those around them if not properly decontaminated. Radiation exposure initially includes gamma and neutron exposure in the minutes following detonation, leading to acute radiation syndrome (ARS). Over time, a delayed response due to fallout produced by fission products and neutron-induced radionuclides in the surrounding environment is also a concern. The risks related to fallout are highest in the hours or days immediately following detonation (REMM, 2021c). Radiation exposure is expressed in gray (Gy), the international system unit of radiation dose. 1 Gy equals 1 joule/kg and is equivalent to 100 rad (the older designation). Gy does not describe the biological effect of a particular type of radiation, which has varying quality or weighting factors (REMM, 2021b).
There are zones following a nuclear emergency: damage zones (severe, moderate, and light) and fallout zones. The severe damage zone is defined as within 0.5 miles of the detonation site. Buildings are destroyed, and no emergency responders enter for at least 72 hours (pending instructions). Survivors are not likely within this zone. The moderate damage zone is within 0.5-1 mile from the detonation site, with significant building damage. Visibility will be poor in this area with downed power lines, rubble, and overturned vehicles. Injuries in this area will be severe, representing the best lifesaving opportunities. The light damage zone is 1-3 miles from the detonation site, with broken windows and rarely life-threatening injuries. Fallout is defined as the spread of radioactive contamination from the detonation site. This zone changes with time and wind/weather conditions. The greatest danger from fallout is within 20 miles from the detonation site, as radioactive dust may cause up to 0.1 Gy per hour of radiation exposure. This danger decreases as the radioactivity decays. Activity in the fallout zone should be planned and minimized as much as possible. Survivors within the fallout zone should be instructed to shelter in place. Cumulative radiation exposure (0.0001 to 0.1 Gy/hour) may extend hundreds of miles from the detonation site (US Government Interagency Report, 2016).
In addition to the radiation exposure, blast injuries from a nuclear detonation or improvised nuclear device (IND) result from barotrauma affecting air-filled organs and air-fluid interfaces. At 5 pounds per square inch (psi), this can rupture a patient’s tympanic membrane, cause lung injury at 15 psi, or cause fatal injuries to hollow viscera at 50 psi. Secondary blast injuries may include penetrating trauma and fragmentation. Tertiary blast injuries might consist of skeletal fractures, blunt trauma, or traumatic brain injuries (TBI). Finally, quarternary injuries include burns, asphyxia, and toxic inhalants. In addition, thermal energy released during a detonation may cause widespread fires, leading to extensive burns and eye injuries (e.g., flash blindness, retinal scarring) in addition to widespread property damage and difficulty accessing or utilizing existing medical facilities (REMM, 2021c).
First receivers caring for patients following such an event should be wearing a personal radiation dosimeter to track their exposure. Those functioning in the HDZ should wear level C PPE described in Table 4, including a full-face air-purifying respirator (with a p-100 or HEPA filter). Level D PPE is likely sufficient for those HCPs working in the post-decontamination zone, where patients may be internally contaminated but not externally. Lead aprons are ineffective at protecting against high-energy, highly penetrating ionizing radiation, in addition to being cumbersome. Pregnant HCPs should not be working in the HDZ, focusing instead on areas with reduced environmental exposure and caring for those who are not internally contaminated. A hard hat, coated hood, or helmet may be indicated for HCPs working in the field to protect against falling debris. Coated coveralls should be worn if functioning in the HDZ. Ear protection prevents dust particles from entering the ear canal. Disposable nitrile or neoprene gloves are acceptable, and latex booties over sturdy footwear or neoprene boots (REMM, 2021c; US Government Interagency Report, 2016).
Decontamination should begin with clothing removal, which can reduce external contamination by as much as 90%. Clothing and shoes should be placed into a sealed and properly labeled bag. Patients can be scanned using a radiation survey meter for external contamination. A whole-body radiation scan should be performed at baseline, if possible, followed by two cycles of whole-body decontamination. The goal should be to reduce external contamination to less than 2x background levels. Tepid water and a mild, neutral pH cleanser should be used for decontamination. Hair should be washed using shampoo, but conditioner should be avoided as it binds proteins. This should be followed by the removal and potentially the debridement of radioactive shrapnel. Surrounding skin should be covered, and open wounds irrigated. Long surgical instruments should be used to remove any visible foreign bodies. Injuries may require surgical debridement if survey meter readings remain elevated. Entrance sites (i.e., ears, nose, and mouth) should be swabbed and surveyed to assess for contamination. An ear syringe may be used to irrigate the external ear canal after confirming an intact tympanic membrane. Teeth should be brushed and the mouth rinsed with 3% hydrogen peroxide to reduce mouth contamination. Eyes may be irrigated using sterile water or saline if the globe is intact only, directing runoff towards the outer canthus. All towels, etc., should be placed into a sealed and labeled bag as clothing above. This process may need to be modified based on the supply and demand of available resources in a mass casualty event (REMM, 2021c).
Acute Radiation Syndrome Diagnosis and Management
Acute radiation syndrome (ARS) is typically four subsyndromes (i.e., hematopoietic, GI, cutaneous, and neurovascular) of varying intensity that progress through four phases, depending on the amount of radiation exposure (REMM, 2021a):
- The hematopoietic subsyndrome of ARS is characterized by decreased granulocyte WBCs (e.g., neutrophils, basophils, and eosinophils), lymphopenia, and thrombocytopenia. Transfusion of blood products may be required, typically 2-4 weeks after radiation exposure. Cellular blood products should be irradiated before transfusion using 25 Gy of external beam radiation to prevent graft versus host disease (GVHD). These products should also be leukocyte reduced to avoid the risk of post-transfusion fevers, immunosuppression, platelet alloimmunization, and cytomegalovirus infection. These protective measures may not be possible in a mass casualty event scenario.
- Platelet transfusions are recommended for platelet counts below 10,000/µL (10 x 109/L) if the patient is closely observed and has no active bleeding or other complications. This threshold should be increased to encourage transfusion for platelet counts below 20,000/µL (20 x 109/L) if the patient cannot be closely observed, is actively bleeding, or is at increased risk of bleeding. In those patients with trauma, surgery, or cerebral edema, transfusion of platelets should be considered at 50,000/µL (50 x 109/L) or below if resources allow.
- Romiplostim (NPlate) is a thrombopoietin receptor agonist which increases platelet production by activating the thrombopoietin receptor.
- Packed red blood cell (PRBC) transfusion should be considered for patients with a hemoglobin level below 10 g/dL.
- A myeloid colony-stimulating factor (CSF) may be considered if 2 Gy or more of exposure to the whole or a significant portion of the body. A baseline CBC with differential should be obtained before therapy. These medications may cause bone pain, splenic enlargement or rupture, acute respiratory distress syndrome, allergic reactions, sickle cell crisis, alveolar hemorrhage with hemoptysis, capillary leakage, thrombocytopenia, or leukocytosis. Currently, there are three FDA-approved CSF agents.
- Filgrastim (Neupogen) is a granulocyte-CSF (G-CSF) that should be dosed at 10 µg/kg/day and administered SQ daily. If resources allow, CBC should be checked daily in these patients. Pediatric patients may be dosed at 5 µg/kg/day.
- Pegfilgrastim (Neulasta) is a pegylated G-CSF. Patients should receive 6 mg SQ weekly for two doses if they are at least 45 kg. Patients under 45 kg should be dosed based on weight per the manufacturer’s instructions.
- Sargramostim (Leukine) is a granulocyte-macrophage CSF that should be dosed at 7 µg/kg/day and administered SQ daily in patients that are at least 40 kg. Patients below 40 kg should be dosed at 10-12 µg/kg/day. If resources allow, CBC should be checked daily in these patients.
- Platelet transfusions are recommended for platelet counts below 10,000/µL (10 x 109/L) if the patient is closely observed and has no active bleeding or other complications. This threshold should be increased to encourage transfusion for platelet counts below 20,000/µL (20 x 109/L) if the patient cannot be closely observed, is actively bleeding, or is at increased risk of bleeding. In those patients with trauma, surgery, or cerebral edema, transfusion of platelets should be considered at 50,000/µL (50 x 109/L) or below if resources allow.
- The GI subsyndrome of ARDS is characterized by N/V, the severity and onset of which correlate well with the radiation dose exposure. The REMM website offers HCPs a biodosimetry tool that calculates the estimated exposure for a patient based on this crucial data. Patients will often also develop diarrhea, abdominal pain and cramping, mucosal loss, and GI bleeding.
- According to McKenzie and colleagues (2019), the use of a 5-HT3 (serotonin) receptor antagonist (e.g., ondansetron [Zofran], granisetron [Sustol], dolasetron [Anzemet], and palonosetron [Aloxi]) may help to reduce N/V.
- Loperamide (Lomotil) can be used to help reduce diarrhea severity.
- The patient’s daily weight and intake/output should be monitored closely with IV fluids and electrolytes to replace losses and maintain balance.
- The cutaneous subsyndrome of ARS appears hours (and sometimes weeks) following the exposure. It is characterized initially by erythema, edema, blistering, skin sensitivity, and itching. Then, days to weeks after exposure, the patient may present with desquamation (wet to dry), hair loss, onycholysis (nail and nail bed changes), and ulceration or necrosis.
- Appropriate wound care with or without debridement is crucial for patient comfort and infection control purposes.
- Water-based topical corticosteroid ointments may be used to decrease inflammation.
- Topical antimicrobials are preferred over systemic agents for antimicrobial prophylaxis.
- Nonadherent dressings should be applied to open wounds or blisters and changed regularly per institutional protocol or as resources allow.
- Fluid replacement and pain management should also be considered nursing priorities in these patients.
- Aqueous lotions and mild soaps may be soothing.
- Topical or systemic antihistamines may alleviate itching.
- Appropriate wound care with or without debridement is crucial for patient comfort and infection control purposes.
- The neurovascular subsyndrome of ARS is characterized initially (within hours) by the onset of N/V and changes in the patient’s blood pressure and body temperature. Within days to weeks, this subsyndrome may also include fatigue, headache, anorexia, and cognitive deficits.
- Diligent and consistent neurological exams are paramount to the nursing care of radiation victims. In addition, the patient’s daily weight and intake/output should be monitored closely with IV fluids or pressors helpful in those who develop hypotension.
- Fluid restrictions may be required in those who develop increased intracranial pressure (ICP). In those with severely elevated ICP or shock, systemic corticosteroids or mannitol (Osmitrol) may be required.
- Acetaminophen (Tylenol) should be used as an antipyretic if indicated.
The four stages of ARS include the prodromal, latent, manifest illness, and recovery or death phases (REMM, 2021b; US Government Interagency Report, 2016):
- If exposed to 1-2 Gy, a patient typically experiences the prodromal phase for at least 2 hours, with 10-50% of victims vomiting in 3-6 hours and lasting less than 24 hours. The latent phase extends through days 21 to 35, and manifest illness typically begins at least 30 days after initial exposure. Recovery is expected unless the patient’s course is complicated by trauma or pre-existing chronic conditions. Symptoms are primarily within the hematopoietic subsyndrome in these patients, with no CNS impairment. A documented long-term effect, there is an 8-16% increased lifetime risk of developing cancer after 1-2 Gy of radiation exposure.
- If exposed to 2-4 Gy, most patients experience hematopoietic symptoms and some GI. The prodromal phase lasts for 1-2 hours. 70-90% of victims start vomiting in 2-4 hours, lasting less than 24 hours. 10-80% of clients are febrile. Cognitive impairment may be present for 6-20 hours. The lymphocyte count typically drops below 1,000 cells/µL at 24 hours. The latent phase lasts through days 18 to 28, after which the manifest illness phase begins. This third phase can last for weeks to months, varying with GI and bone marrow recovery. Death is “potentially avoidable” in these patients with proper supportive care. The LD 50/60 (lethal dose of radiation with no medical intervention to 50% of the population at day 60) is between 3.2-4.5 Gy. There is a 24% increased lifetime risk of developing cancer after 3 Gy of radiation exposure.
- In victims exposed to 4-6 Gy, hematopoietic symptoms are severe, GI symptoms are moderate, and some neurovascular symptoms are expected. The prodromal phase lasts less than 1 hour following exposure, with 100% of victims vomiting in 2-4 hours and lasting less than 24 hours. 80-100% of victims develop a fever within 2 hours, and the lymphocyte count typically drops below 1,000 cells/µL by 24 hours. Cognitive impairment may be present for 6-20 hours. The latency phase then continues for 8 to 18 days, after which manifest illness may last for weeks to months. The recovery phase may take months to years in this cohort. Moderate to complete hair loss is expected, and a 20-70% mortality rate can be expected at this dose, typically during weeks 4-8. There is > 40% increased lifetime risk of developing cancer after 6 Gy of radiation exposure.
- After exposures of 6-8 Gy, hematopoietic and GI symptoms are severe, while neurovascular symptoms are moderate. The prodromal phase lasts less than 30 minutes, with 100% of victims vomiting within 1-2 hours and lasting less than 48 hours. All victims are febrile; 80% develop a severe headache within 3-4 hours and often a decreased level of consciousness. Cognitive impairment may be present for greater than 20 hours. The lymphocyte count typically drops below 500 cells/µL at 24 hours. The latency phase lasts less than 7 days in these patients, at which point manifest illness begins, lasting for months. Complete hair loss is expected by day 11. Recovery, if it occurs, is expected to take years. These patients require urgent hospitalization. HCPs should expect a mortality rate above 50% at this exposure level beginning at 1-2 weeks.
- If exposed to 8 Gy or more, the prodromal phase typically lasts less than 10 minutes, and the latency phase is often nonexistent. All subsyndromal symptoms are severe. 100% of patients experience vomiting within an hour and lasting less than 48 hours. All victims experience diarrhea and fever, while 80-90% will report a severe headache or loss of consciousness for seconds to minutes. There is rapid cognitive decline. The lymphocyte count drops within hours. Manifest illness begins almost immediately, with profuse, bloody diarrhea by days 4 or 5 and complete hair loss by day 10. Seizures are common. Most of these patients are appropriate for palliative-level care, with a select few surviving with aggressive supportive care.
References
Bhalla, M. C., Frey, J., Rider, C., Nord, M., & Hegerhorst, M. (2015). Simple triage algorithm and rapid treatment and sort, assess, lifesaving, interventions, treatment, and transportation mass casualty triage methods for sensitivity, specificity, and predictive values. The American Journal of Emergency Medicine, 33(11), 1687–1691. https://doi.org/10.1016/j.ajem.2015.08.021
Bower, W. A., Hendricks, K., Pillai, S., Guarnizo, J., & Meaney-Delman, D. (2015). Clinical framework and medical countermeasure use during an anthrax mass-casualty incident. Morbidity and Mortality Weekly Report Recommendations and Reports, 64(No. RR-4). https://www.cdc.gov/mmwr/pdf/rr/rr6404.pdf
California Department of Public Health. (2021). Infant botulism treatment and prevention program. https://www.cdph.ca.gov/Programs/CID/DCDC/Pages/ObtainBabyBig.aspx
Centers for Disease Control and Prevention. (2016a). Anthrax vaccine recommendations. https://www.cdc.gov/vaccines/vpd/anthrax/hcp/recommendations.html
Centers for Disease Control and Prevention. (2016b). Melioidosis for healthcare workers. https://www.cdc.gov/melioidosis/health-care-workers/index.html
Centers for Disease Control and Prevention. (2016c). Standard precautions for all patient care. https://www.cdc.gov/infectioncontrol/basics/standard-precautions.html
Centers for Disease Control and Prevention. (2017a). Glanders bioterrorism response planning. https://www.cdc.gov/glanders/bioterrorism-response-planning/index.html
Centers for Disease Control and Prevention. (2017b). Smallpox vaccination information for clinicians. https://www.cdc.gov/smallpox/clinicians/vaccination.html
Centers for Disease Control and Prevention. (2018a). Bioterrorism agents and diseases by category. https://emergency.cdc.gov/agent/agentlist-category.asp
Centers for Disease Control and Prevention. (2018b). Clinician outreach and communication activity (COCA). https://emergency.cdc.gov/coca/
Centers for Disease Control and Prevention. (2018c). For US healthcare settings: Donning and doffing PPE for evaluating PUIs for Ebola who are clinically stable and do not have bleeding, vomiting, or diarrhea. https://www.cdc.gov/vhf/ebola/healthcare-us/ppe/guidance-clinically-stable-puis.html
Centers for Disease Control and Prevention. (2018d). Guidance on personal protective equipment (PPE) to be used by healthcare workers during management of patients with confirmed Ebola or persons under investigation (PUIs) for Ebola who are clinically unstable or have bleeding, vomiting, or diarrhea in US hospitals, including procedures for donning and doffing PPE. https://www.cdc.gov/vhf/ebola/healthcare-us/ppe/guidance.html
Centers for Disease Control and Prevention. (2018e). Ricin: Information and guidance for clinicians. https://emergency.cdc.gov/agent/ricin/clinicians/index.asp
Centers for Disease Control and Prevention. (2019a). Botulism. https://www.cdc.gov/botulism/index.html
Centers for Disease Control and Prevention. (2019b). Lassa fever. https://www.cdc.gov/vhf/lassa/index.html
Centers for Disease Control and Prevention. (2019c). Smallpox for clinicians. https://www.cdc.gov/smallpox/clinicians/index.html
Centers for Disease Control and Prevention. (2020a). Anthrax information for healthcare professionals. https://www.cdc.gov/anthrax/healthcare/index.html
Centers for Disease Control and Prevention. (2020b). The health alert network. https://emergency.cdc.gov/HAN/
Centers for Disease Control and Prevention. (2020c). The CDC healthcare preparedness toolbox. https://www.cdc.gov/cpr/readiness/healthcare/toolbox.htm
Centers for Disease Control and Prevention. (2020d). Plague: Resources for clinicians. https://www.cdc.gov/plague/healthcare/clinicians.html
Centers for Disease Control and Prevention. (2021a). Ebola (Ebola virus disease) for clinicians. https://www.cdc.gov/vhf/ebola/clinicians/index.html
Centers for Disease Control and Prevention. (2021b). The national syndromic surveillance program. https://www.cdc.gov/nssp/overview.html
Centers for Disease Control and Prevention. (2021c). Tularemia for clinicians. https://www.cdc.gov/tularemia/clinicians/index.html
Chemical Hazards Emergency Medical Management. (2021a). CHEMM intelligent syndromes tool. https://chemm.nlm.nih.gov/chemmist.htm
Chemical Hazards Emergency Medical Management. (2021b). Information for hospital providers. https://chemm.nlm.nih.gov/hospitalproviders.htm
Chemical Hazards Emergency Medical Management. (2021c). Nerve agents: Emergency department and hospital management. https://chemm.nlm.nih.gov/na_hospital_mmg.htm#top
Chemical Hazards Emergency Medical Management. (2021d). Personal protective equipment. https://chemm.nlm.nih.gov/ppe.htm
Hendricks, K. A., Wright, M. E., Shadomy, S. V., Bradley, J. S., Morrow, M. G., Pavia, A. T., Rubinstein, E., Holty, J. E. C, Messonnier, N. E., Smith, T. L., Pesik, N., Treadwell, T. A., Bower, W. A., & The Workgroup on Anthrax Clinical Guidelines. (2014). Centers for Disease Control and Prevention expert panel meetings on prevention and treatment of anthrax in adults. Emerging Infectious Diseases, 20(2). http://dx.doi.org/10.3201/eid2002.130687
Hick, J. L., Barbera, J. A., & Kelen, G. D. (2009). Refining surge capacity: Conventional, contingency, and crisis capacity. Disaster Medicine and Public Health Preparedness, 3(2 Suppl), S59–S67. https://doi.org/10.1097/DMP.0b013e31819f1ae2
Institute of Medicine. (2012). Crisis standards of care: A systems framework for catastrophic disaster response. National Academies Press (US). https://doi.org/10.17226/13351
Kosnett, M. J. (2013). The role of chelation in the treatment of arsenic and mercury poisoning. Journal of Medical Toxicology: Official Journal of the American College of Medical Toxicology, 9(4), 347–354. https://doi.org/10.1007/s13181-013-0344-5
Lipsitz, R., Garges, S., Aurigemma, R., Baccam, P., Blaney, D. D., Cheng, A. C., Currie, B. J., Dance, D., Gee, J. E., Larsen, J., Limmathurotsakul, D., Morrow, M. G., Norton, R., O'Mara, E., Peacock, S. J., Pesik, N., Rogers, L. P., Schweizer, H. P., Steinmetz, I., Tan, G., … Smith, T. L. (2012). Workshop on treatment of and postexposure prophylaxis for Burkholderia pseudomallei and B. mallei infection, 2010. Emerging Infectious Diseases, 18(12), e2. https://doi.org/10.3201/eid1812.120638
McKenzie, E., Zaki, P., Raman, S., Olson, R., McFarlane, T., DeAngelis, C., Chan, S., Pidduck, W., Razvi, Y., Bushehri, A., & Chow, E. (2019). Radiation-induced nausea and vomiting: A comparison between MASCC/ESMO, ASCO, and NCCN antiemetic guidelines. Supportive Care in Cancer: Official Journal of the Multinational Association of Supportive Care in Cancer, 27(3), 783–791. https://doi.org/10.1007/s00520-018-4586-2
Medscape. (n.d.). Ribavirin. Retrieved July 9, 2021, from https://reference.medscape.com/drug/rebetol-ribasphere-ribavirin-342625
National Academy of Sciences. (2004). Chemical attack: Warfare agents, industrial chemicals, and toxins. News & Terrorism: Communicating in a Crisis. https://www.dhs.gov/xlibrary/assets/prep_chemical_fact_sheet.pdf
Pillai, S. K., Huang, E., Guarnizo, J. T., Hoyle, J. D., Katharios-Lanwermeyer, S., Turski, T. K., Bower, W. A., Hendricks, K. A., & Meaney-Delman, D. (2015). Antimicrobial treatment for systemic anthrax: Analysis of cases from 1945 to 2014 identified through a systematic literature review. Health Security, 13(6), 355–364. https://doi.org/10.1089/hs.2015.0033
Radiation Emergency Medical Management. (2021a). Managing acute radiation syndrome (ARS). https://www.remm.nlm.gov/ars.htm#
Radiation Emergency Medical Management. (2021b). Time phases of acute radiation syndrome (ARS). https://www.remm.nlm.gov/ars_timephases1.htm
Radiation Emergency Medical Management. (2021c). Top 10 Items to know for radiation emergencies. https://www.remm.nlm.gov/wheretostart.htm
Rao, A. K., Sobel, J., Chatham-Stephens, K., & Luquez, C. (2021) Clinical guidelines for diagnosis and treatment of botulism. Morbidity and Mortality Weekly Report Recommendations and Reports, 70(No. RR-2), 1–30. http://dx.doi.org/10.15585/mmwr.rr7002a1
Ready.gov. (2021). Bioterrorism. https://www.ready.gov/Bioterrorism
Siegel, J. D., Rhinehart, E., Jackson, M., Chiarello, L., and the Healthcare Infection Control Practices Advisory Committee. (2007). Guideline for isolation precautions: Preventing transmission of infectious agents in healthcare settings. https://www.cdc.gov/infectioncontrol/guidelines/isolation/index.html
US Federal Bureau of Investigation (n.d.-a). Terrorism. Retrieved July 6, 2021, from https://www.fbi.gov/investigate/terrorism
US Federal Bureau of Investigation (n.d.-b). Weapons of mass destruction. Retrieved July 6, 2021, from https://www.fbi.gov/investigate/wmd
US Food and Drug Administration. (2018). BAT (botulism antitoxin heptavalent [A, B, C, D, E, F G]-equine. https://www.fda.gov/vaccines-blood-biologics/approved-blood-products/bat-botulism-antitoxin-heptavalent-b-c-d-e-f-g-equine
US Food and Drug Administration. (2020a). FDA approves first treatment for Ebola Virus. https://www.fda.gov/news-events/press-announcements/fda-approves-first-treatment-ebola-virus
US Food and Drug Administration. (2020b). FDA approves treatment for Ebola virus. https://www.fda.gov/drugs/drug-safety-and-availability/fda-approves-treatment-ebola-virus
US Government Interagency Report. (2016). Quick reference guide: Radiation risk information for responders following a nuclear detonation. https://www.dhs.gov/sites/default/files/publications/Quick%20Reference%20Guide%20Final.pdf
US Occupational Safety and Health Administration. (2005). OSHA best practices for hospital-based first receivers of victims from mass casualty incidents involving the release of hazardous substances. https://www.osha.gov/sites/default/files/firstreceivers_hospital.pdf